|
Name the electron-pair geometry and mo- lecular geometry for each 1. (Valence electrons are the number of electrons present in the outermost shell of an atom). Predict the hybridization, geometry, and bond angles for the central atoms in but-2-ene, CH3CHCHCH3 CH3CHNH Determine the electron geometry, molecular geometry, and hybridization for both carbons in CH3COOH. In order to draw the lewis structure of CF4, first of all you have to find the total number of valence electrons present in the CF4 molecule. Determine the name for the following molecular geometry and the ideal bond angle(s). 
Here, the given molecule is CF4 (carbon tetrafluoride). In the case of bond formation in Acetylene molecule: Name the type of Hybridization. Lewis structure & Electronic geometry & Molecular geometry hline a) CF4. Step 1: Calculate the total number of valence electrons. 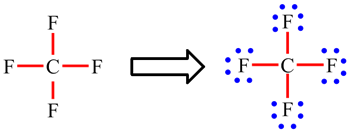
(b) The molecular structure is square planar with the lone pairs directly across from one another. Figure 7.25 (a) XeF4 adopts an octahedral arrangement with two lone pairs (red lines) and four bonds in the electron-pair geometry. In CF4, the tetrahedral arrangement of the fluorine atoms around the central carbon atom leads to a symmetrical charge distribution, resulting in a nonpolar molecule. The molecular geometry of CF4 and SF4 plays a crucial role in determining their polarity. For a trigonal planar structure, the bond angle is 120°. The five atoms are all in the same plane and have a square planar molecular structure. Analysis of the Molecular Geometry and Electronegativity in CF4 and SF4. 
The molecular geometry is the same as the electron geometry. The shape is not distorted because there are no lone pairs on the central boron atom. The electron geometry of BF 3 is trigonal planar. These structures can generally be predicted, when A is a nonmetal, using the "valence-shell electron-pair repulsion model (VSEPR) discussed in the next section. Explain geometry of methane molecule on the basis of Hybridization. These three hybrid orbitals overlap with fluorine’s 2p orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
